 
These have half lives of 50 days and 30 years, respectively. There are also reports of strontium-89 and strontium-90 being discovered in the environment near the plant. "It's a testimony to the sensitivity of radiation detection equipment that the plutonium can be detected and its source distinguished from such tiny quantities, not a cause for alarm." "You can find equivalent levels in soil around the world dispersed from atmospheric bomb tests decades ago." Plutonium releases mostly alpha radiation, and is a serious health hazard if ingested or inhaled in significant amounts, remaining in the lungs, or absorbed into the blood stream where it moves into bones, liver or other organs exposing surrounding tissue to radiation for decades.īut plutonium levels detected in soil near the Fukushima plant are "tiny", says Crossley. As well as being a by-product of nuclear reactions, it's also part of the fuel mix used at the power station's number three reactor. The Japanese government has confirmed three plutonium isotopes Pu-238, Pu-239 and Pu-240, have been detected in soil in the exclusion zone near the Fukushima facility. "That's why there are exclusion zones in place." The risk of radiation poisoning following the Fukushima incident will be related to the level of exposure people face, says Crossley. Just how long is indicated by the isotope's 'half-life' - the time it takes for half of a radioactive isotope to decay away. Some radioactive substances can remain dangerous in the environment longer than others. Plutonium and uranium produce a range of radioactive fission products including variations of iodine, caesium, strontium and noble gases such as argon and xenon. "It works exactly the same as lower doses of radiation but on a mass scale and when enough cells die that can cause problems", says Crossley. As the radiation dose gets higher (many sieverts) this can kill you very quickly, in a matter of hours if the dose is high enough." "This often appears as skin burns and gastrointestinal effects. "Higher doses (over a sievert) can cause immediate health problems because a larger number of cells are killed off by the radiation." DNA can be repaired, but if the repair is incorrect, it could cause mutation which could lead to cancer later on". "Radiation damages cells by breaking chromosomes in DNA. "Contamination is when you get the radioactive material itself on you or in you, including eating it breathing it in or absorbing it through the skin." "You don't become radioactive from external irradiation", says Crossley. But it only lasts as long as you're near the source, once you move away, you are not being irradiated any more." "External irradiation is when you absorb the energy of radiation which can be in the form of beta, alpha or neutron particles or as gamma and/or X-rays. Gamma or x-rays, which are electromagnetic radiation similar to light, but with much more energy.įind out more about radiation in our Bernie's Basics: It's radioactive man.Īccording to Crossley, there are two ways you can be exposed to radiation. Neutron radiation - consisting of free neutrons which are released during fission but not usually through radioactive decay. Types of ionising radiation include:Īlpha particles - two protons and two neutrons bound together like a helium nucleus - are large particles that can do lots of damage, but only over short distances.īeta particles - are electrons, which can travel longer distances in air and tissue than alpha particles. It can cause damage by breaking chemical bonds. What is ionising radiation?Ionising radiation has enough energy to remove electrons from atoms. 
The bits of smashed atom (fission fragments) are unstable isotopes that become stable by releasing energy as ionising radiation. In a reactor when neutrons are smashed into an atom, the atom releases neutrons which then hit other atoms in a chain reaction (find out how nuclear reactors work). The amount of radiation harmful to people depends on which chemical element, and how much of it they are exposed to, says Steven Crossley, a radiation physicist at Sir Charles Gairdner Hospital in Western Australia.ĭifferent variations of elements, known as isotopes, decay by different methods and give off different types of radiation, so they can have different effects on the body, he says. 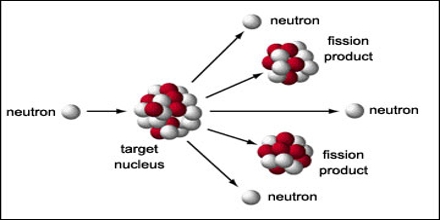
Over the last few weeks, "above-legal" levels of radioactive iodine-131, caesium-134 and caesium-137 have been detected in marine life off the coast of Japan.Īnd more recently, plutonium and strontium-89 and strontium-90 have been detected in the soil around the damaged Fukushima nuclear plant. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
